 ĭensity of crude oil, Density of fuel oils, Density of lubricating oil and Density of jet fuel as function of temperature.Īs shown in the figures, the change in density is not linear with temperature - this means that the volumetric expansion coefficient for water is not constant over the temperature range. įor other substances, see density and specific weight of acetone, air, ammonia, argon, benzene, butane, carbon dioxide, carbon monoxide, ethane, ethanol, ethylene, helium, hydrogen, methane, methanol, nitrogen, oxygen, pentane, propane and toluene. See also other properties of Water at varying temperature and pressure : Boiling points at high pressure, Boiling points at vacuum pressure, Dynamic and kinematic viscosity, Enthalpy and entropy, Heat of vaporization, Ionization Constant, pK w, of normal and heavy water, Melting points at high pressure, Prandtl number, Properties at Gas-Liquid Equilibrium Conditions, Saturation pressure, Specific gravity, Specific heat (heat capacity), Specific volume, Thermal conductivity, Thermal diffusivity and Vapour pressure at gas-liquid equilibrium. See Water and Heavy Water for thermodynamic properties at standard condtions. The density of water depends on temperature and pressure as shown below: water table and also somewhere near -60 ft at the top of a permeable zone in. At 20 degrees Celsius it is 998. It is exactly 1000 kg/m3 at 4 degrees Celsius. But you’re right that it does vary a little bit with temperature. Note! Temperature must be within the ranges 0-370 ☌, 32-700 ☏, 273-645 K and 492-1160 °R to get valid values. In general, we say that the density of water is 1000 kg/m3 (or 1 g/cm3). The output density is given as g/cm 3, kg/m 3, lb/ft 3, lb/gal(US liq) and sl/ft 3. The calculator below can be used to calculate the liquid water density at given temperatures. The density of water at 20 degrees Celsius is 998.20 kilograms per cubic meter or 0.9982 grams per cubic centimeter. 
See more about the difference between mass and weight Online Water density Calculator The density of water is 1.940 sl/ft 3at 39 ☏ (4 ☌), and the specific weight in Imperial units is In the Imperial system the mass unit is the slug, and is derived from the pound-force by defining it as the mass that will accelerate at 1 foot per square second when a 1 pound-force acts upon it :ġ = 1 * 1 and 1 = 1 /1 In the SI system, specific weight of water at 4☌ will be: G = acceleration due to gravity, units typically and value on Earth usually given as 9.80665 m/s 2or 32.17405 ft/s 2 Specific weight is the ratio of the weight to the volume of a substance: Company About Us Responsibility Events Press Releases Programs Careers Offices. The density of water (H 2 O) changes depending on temperature and pressure - as with other substances. Pure water has its highest density 1000 kg/m 3 or 1.940 slug/ft 3 at temperature 4☌ (=39.2☏). DENWAT Pure Water Density Standard a certified reference material to BSEN ISO / IEC 17025 is used for calibrating instruments for measuring density. This is the typical heat capacity of water.Density is the ratio of the mass to the volume of a substance: For example, say that we want to reduce the sample's thermal energy by 63,000 J. If you want to cool down the sample, insert the subtracted energy as a negative value. The output density is given as g/cm, kg/m, lb/ft, lb/gal (US liq) and sl/ft Temperature must be within the ranges 0-370 C, 32-700 F, 273-645 K and 492-1160 R to get valid values. Density of atmospheric water vapor was computed using (Brutsaert, 1982. Online calculator with Saturated Steam Table by Temperature. See more about the difference between The calculator below can be used to calculate the liquid water density at given temperatures. degrees Celsius, to convert it to kelvins and other terms are as defined previously. Steam Bulletin: Archive - Email Magazine. 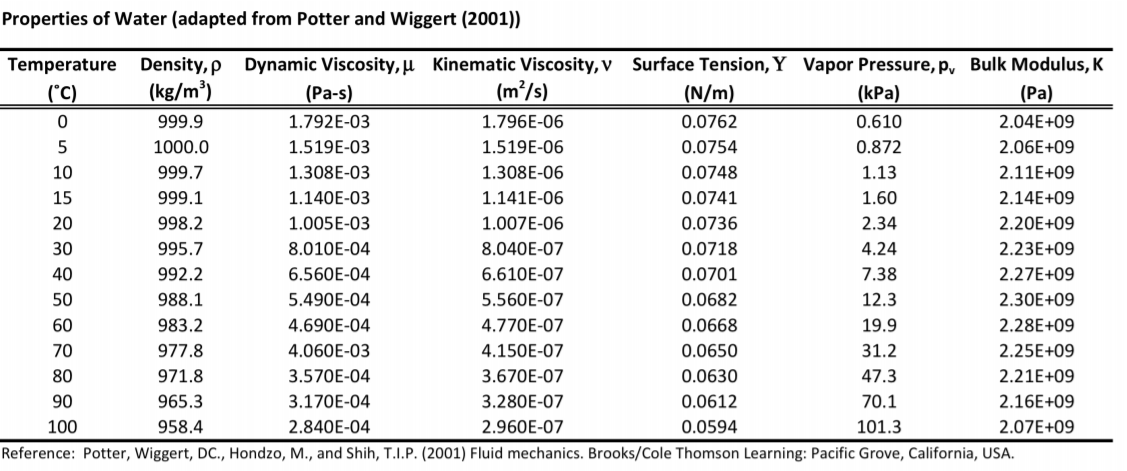 
Insert the amount of energy supplied as a positive value. TLV ToolBox - Mobile App for Steam Engineering. Determine whether you want to warm up the sample (give it some thermal energy) or cool it down (take some thermal energy away). 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
